Distinguished Research Project at KFUPM : Converting Water into a Source of Clean Energy (H2)
Dec. 20, 2018 | ResearchHydrogen (H2) is required for several petrochemical processes, and the worth of world H2 market is expected to exceed $180 billion by 2024. In 2015, the H2 market share of Saudi Arabia was > $3 billion, which is likely to grow another ~ 5% by 2024. Currently, most of the H2 is produced from methane present in the natural gas – the process is called ‘steam–methane reforming’. However, this process is energy-intensive, carried out at 700 °C–1000 °C, consumes fossil fuel and, more importantly, adds to carbon dioxide (CO2) emissions. The development of a sustainable and eco-friendly process, therefore, for large-scale H2 production without CO2 emission would be of great significance for the local industries.
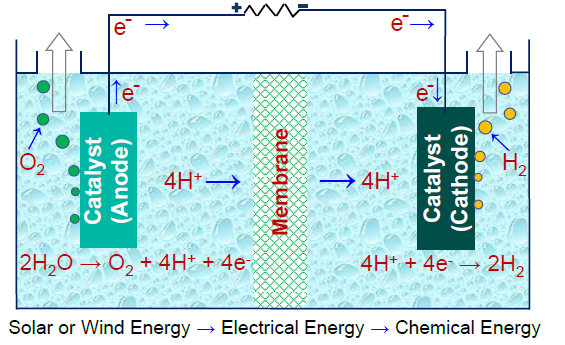
From environmental and energy viewpoints, the production of H2 using electrical energy and water offers great promise. A schematic of the process showing the formation of oxygen and hydrogen gases from water is presented Above. In this process, called electrolysis, renewably-generated surplus electrical energy can be used to break water molecules into hydrogen and oxygen. Firstly, water molecules split at catalyst’s surface (anode), producing electrons (e-), hydrogen ions (H+) and O2 gas. Oxygen gas can be collected at anode, while e- flows through an external electrical circuit and H+ diffuses through membrane towards cathode. At the cathode, reduction of 4H+ takes place producing H2 gas. These gases are pure and can be used without any further purification process. Reactions occurring at anode and cathode are termed as oxygen evolution reaction (OER) and hydrogen evolution reaction (HER), respectively.
The market share of H2 produced through electrolysis is ~7%, which is likely to grow to ~12% by 2024. Reduction of the capital cost and boosting energy efficiency (electricity to hydrogen) continue to be challenging endeavors. These challenges are directly related to the electrocatalysts, and Nanotechnology, a key enabling technology, can be the clue to overcome the challenges and play crucial role in the advancement of electrolysis.
A research team at KFUPM, led by Dr. Mohammad Qamar and consisting of one post-doctoral fellow and two PhD students, is currently working on a distinguished research project which aims to develop cost-effective, stable and high-performance nanostructured electrode materials to be used in electrolysis to produce hydrogen from water, as well as to demonstrate their efficacy at larger scale in a water electrolyzer.
Two years ago (2016), The team devised a Metal Organic-Framework (MOF)-based novel strategy to synthesize highly active and stable electrocatalyst (dimolybdenum carbide, Mo2C) for HER (J. Mater. Chem. A, 2016, 4, pp 16225-16232). They utilized the highly porous texture of Metal Organic Framework as a confined environment to produce Mo2C with uniform distribution on the carbon surface.

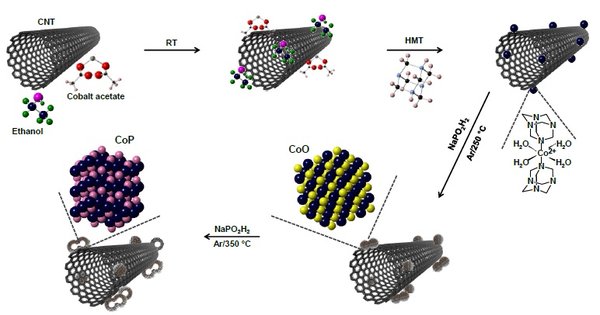
While the as-prepared electrocatalyst showed stable and highly efficient HER, it still required more energy as compared to commercial and benchmark electrocatalyst Pt/C. Recently, they made a significant progress by developing nanostructured electrocatalyst (cobalt monophosphide, CoP) with intriguing features.
The electrocatalyst was grown onto the surface of carbon nanotubes (CNTs). The cost of cobalt (or CoP) is much lower compared to platinum or Pt-based electrocatalysts. Moreover, the preparation method is simple with the potential to scale-up. Hexamethylenetetramine (HMT) is used as a structure-directing agent to guide the formation of interconnected cobalt oxide (CoO), which further grows into interconnected hollow CoP at 350 °C in the presence of argon gas (Ar) and sodium hypophosphite (NaPO2H2).

Endowed with unique attributes, the hybrid electrocatalyst outperformed most of the electrocatalysts reported as yet. In addition, the developed electrocatalyst exhibits outstanding long-term operational stability.
The findings are published in ACS Applied Materials & Interfaces (ACS Appl. Mater. Interfaces, 2018, 10 (35), pp 29407–29416) and documents are prepared to patent the synthesis methodology.
Meanwhile, the team has also developed a low-cost cobalt boride-based novel hybrid electrocatalyst for OER reaction, and the preliminary findings are highly promising.
Furthermore, they are considering the application of such electrocatalysts in other strategically important areas including electrochemical flow technology for converting oil products into highly valuable chemicals, conversion of carbon dioxide (CO2) into value-added chemicals, and energy storage devices (redox flow battery).
All these research are directed to cater the needs of local industries, as well as aligned with Saudi Vision 2030 which envisions reducing Kingdom’s dependence on oil and relying on alternatives diverse economy.

Dr. Mohammad Qamar works as a Research Scientist-II at the Center of Excellence in Nanotechnology (CENT), KFUPM. His current research interest is to develop custom-tailored nanostructured catalysts for energy conversion and storage.
More details of this research in this field could be found at



